Japan approves world’s first regenerative medicines using iPS cells
South China Morning Post
by KyodoFebruary 19, 2026
AI-Generated Deep Dive Summary
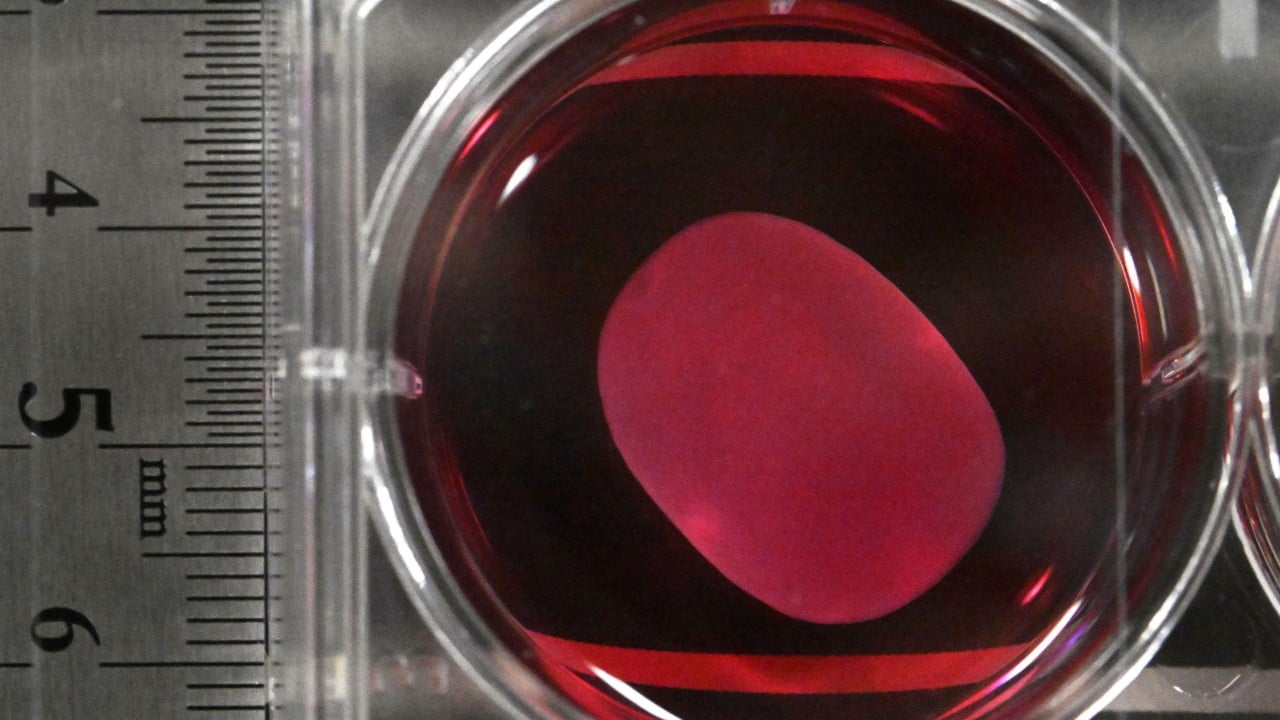
Japan has made a groundbreaking leap in regenerative medicine by becoming the first country to approve treatments using induced pluripotent stem cells (iPS). The nation’s health ministry recently cleared two innovative therapies developed from iPS cells, marking a historic milestone in medical science. These treatments, ReHeart and Amchepry, aim to address severe heart failure and Parkinson’s disease, respectively. ReHeart, created by Cuorips, a startup spun out of Osaka University, uses iPS-derived heart muscle sheets to improve blood vessel formation and restore heart function in patients with ischemic cardiomyopathy. Meanwhile, Amchepry, a collaboration between Sumitomo Pharma and Racthera, targets Parkinson’s disease by generating dopamine neurons from iPS cells to replenish those lost due to the condition.
The approval of these therapies is a testament to over a decade of groundbreaking research led by Shinya Yamanaka, a Nobel Prize-winning scientist. Yamanaka’s discovery of human iPS cells in 2007 revolutionized stem cell science, paving the way for personalized and regenerative treatments. His work has since inspired global advancements in medicine, and this approval represents the first societal application of his pioneering research.
This development is significant not only for Japan but for the entire world. Regenerative medicine holds immense potential to address incurable diseases and reduce
Verticals
worldasia
Originally published on South China Morning Post on 2/19/2026